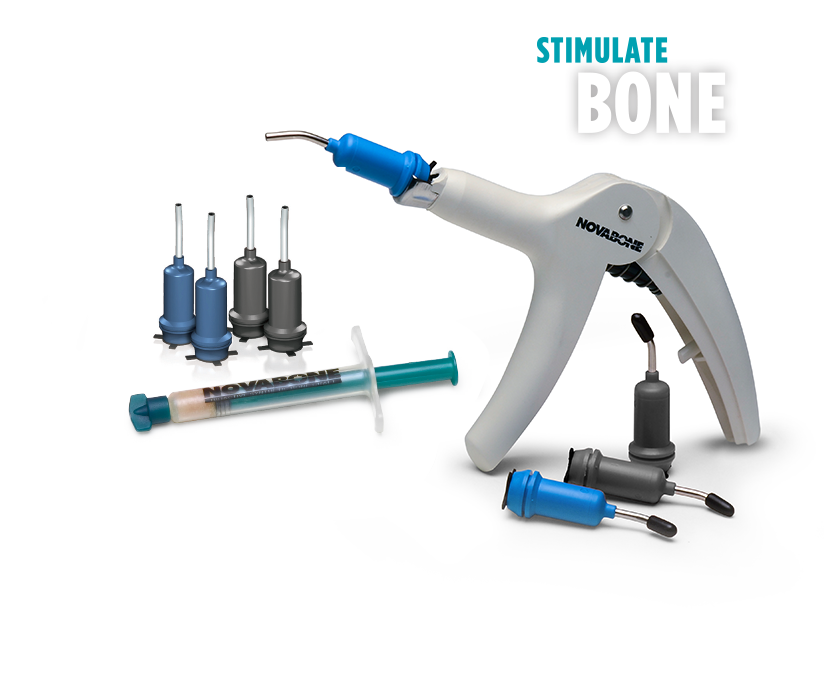
The next generation Calcium-Phosphosilicate bone substitute engineered for enhanced handling and predictable results. NovaBone® Dental Putty is ready to use and extremely user-friendly. It is pre-mixed, cohesive, moldable, and adaptable. NovaBone® Dental Putty is stable at room temperature, does not require refrigeration, has a 4-year shelf-life, and appears radiodense on radiographs.

FEATURES
Unique Formulation
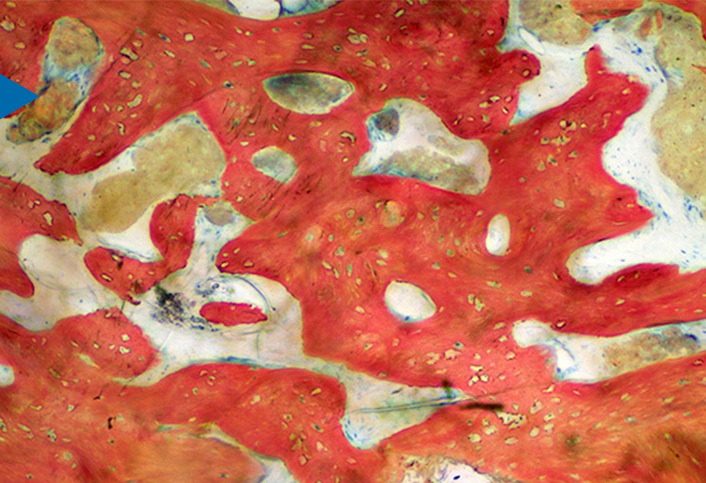
NovaBone® Dental Putty is 100% synthetic and fully resorbable. It is composed of calcium phosphosilicate (CPS) particles in a bimodal size distribution combined with a polyethylene glycol and glycerine binder. The binder improves handling and aids in maintaining the space between the particles, which facilitates revascularization after implantation. The bioactive CPS component makes up 70% of the putty by volume. Upon implantation, the water-soluble binder is absorbed within 24 to 72 hours, creating a 3-dimensional porous scaffold that facilitates diffusion of blood and tissue fluids through the matrix. The smaller CPS particles (32-125 μm) are more rapidly resorbed, providing the initial burst of Ca and P ions. Subsequently, the larger particles (90-710 μm) react, and being more resistant to resorption, continue the process of bone regeneration.
Grafting Simplified
NovaBone® Dental Putty is available in multiple delivery options: trays, pre-filled syringes, and a unique industry-first cartridge delivery system. NovaBone® is the only graft material in the world that is available in disposable uni-dose cartridges. The cartridges simplify dispensing of the graft, especially in hard-to-reach areas, thus facilitating minimally invasive techniques (and hard-to-access defects such as gaps in immediate implant placement and crestal-approach sinus lifts). Cartridges are available in various sizes and are used in conjunction with NovaBone's® cartridge delivery system; each cartridge holds 0.25cc or 0.5cc of putty.
Smart Science™
Unlike most synthetic grafts that are only osteoconductive, bioactive NovaBone® Dental Putty also has an "osteostimulative" effect. After implantation, surface reactions result in absorption of the graft material, a controlled release of Si, Ca, and P ions, and concurrent new bone formation. These surface reactions result in an osteostimulative affect, defined as the stimulation of osteoblast proliferation in vitro as evidenced by increased DNA content and elevated osteocalcin and alkaline phosphatase levels. In vitro gene array analysis has confirmed that when human primary osteoblasts are exposed to extracts of CPS, upregulation of several gene families occurs.
CLINICAL INDICATIONS
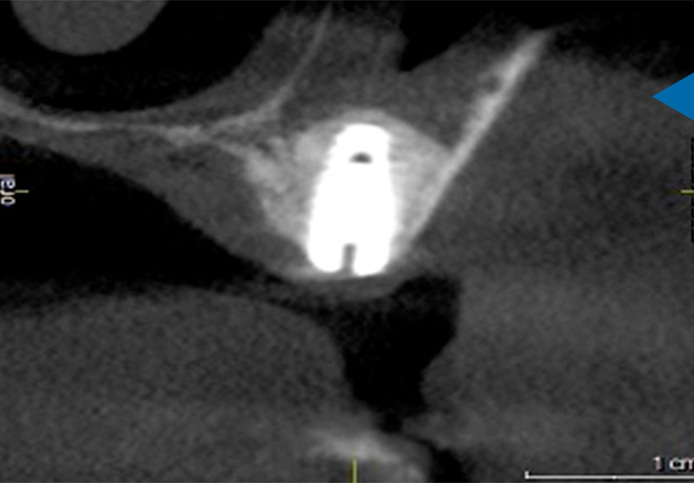
Although approved for use in most indications, NovaBone Dental Putty is best for use in crestal and lateral sinus elevation, immediate implants, partial extraction therapies, socket preservations and periodontal defects.
PRODUCT LITERATURE
Sinus Augmentation
1. A simplified approach to the minimally invasive antral membrane elevation technique utilizing a viscoelastic medium for hydraulic sinus floor elevation. Kotsakis GA, Mazor Z. Oral Maxillofac Surg. 2015; 19(1): 97-101.
2. A clinical and radiographic case series of implants placed with the simplified minimally invasive antral membrane elevation technique in the posterior maxilla. Kher U, Ioannou AL, Kumar T, Siormpas K, Mitsias ME, Mazor Z, Kotsakis GA. J Craniomaxillofac Surg. Dec 2014; 42(8): 1942-7.
3. One-stage transaveolar vs. lateral maxillary sinus augmentation in severely resorbed sites using Calcium Phosphosilicate Putty: A proof of concept study. Kher U, Shanbhag S., Clin. Oral Impl. Res Oct 2014; 25(10).
4. Implants placed simultaneously with lateral-window sinus augmentation utilizing a calcium phosphosilicate putty alloplastic bone substitute for increased primary implant stability: A retrospective study. Udatta Kher, Ziv Mazor, Georgios A. Kotsakis, Panagiotis Stanitsas. Imp Dent 2014, 23(4):496-501.
5. Sinus Elevation with an alloplastic material and simultaneous implant placement: A 1-stage procedure in severely atrophic maxillae. Jodia K, Sadhwani B, Parmar BS, Anchlia S, Sadhwani SB., J Maxillofac. Oral Surg (July-Sept 2014) 13(3):271-280.
6. Minimally Invasive Crestal Approach Technique Utilizing a Cartridge Delivery System. Mazor Z, Ioannou A, Venkataraman N, Kotsakis G, Kher U., Implant Practice. Sept 2013; 6(4): 20-24.
7. Clinical and histologic comparison of two different composite grafts for sinus augmentation: a pilot clinical trial. Galindo-Moreno P, Avila G, Wang HL, et al., Clin Oral Implants Res. Aug 2008; 19(8): 755-9.
Socket Management
1. Clinical Evaluation of 262 Osseointegrated Implants Placed in Sites grafted with Calcium Phosphosilicate Putty: A Retrospective Study. Babbush CA, Kanawati A., J Oral Implantol. Feb 2015; 41(1): 62-9.
2. One-stage transaveolar vs. lateral maxillary sinus augmentation in severely resorbed sites using Calcium Phosphosilicate Putty: A proof of concept study. Kher U, Shanbhag S., Clin. Oral Impl. Res Oct 2014; 25(10).
3. Periotest Values of Implants Placed in Sockets Augmented with Calcium Phosphosilicate Putty Graft: A Comparative Analysis against Implants Placed in Naturally Healed Sockets. Mahesh L, Narayan T, Kostakis G, Shukla S. J Contemp Dent Pract. 2014 Mar 1;15(2):181-5.
4. Alveolar Ridge Preservation with Socket Plug Technique Utilizing an Alloplastic Putty Bone Substitute or a Particulate Xenograft. Mahesh L, Kotsakis G, Venkataraman N, Shukla S, Prasad H., J Oral Implantol. Apr 2015; 41(2): 178-83.
5. A Randomized Blinded Controlled Clinical Study of Particulate Anorganic Bovine Bone Mineral and Calcium Phosphosilicate Putty Bone Substitutes for Socket Preservation. Kotsakis G, Salama M, Chrepa V, Hinrichs JE, Gaillard P., Int. J Oral Maxillofacial Implants. Jan-Feb 2014;29(1):141-51.
6. Histomorphic Evaluation of a Calcium-Phosphosilicate Putty Bone Substitute in Extraction Sockets. Kotsakis G, Joachim F, Saroff S, Mahesh L, Prasad H, Rohrer M. Int J Periodontics Restorative Dent. 2014 Mar-Apr; 34(2)233-9.
7. Histologic and Clinical Evaluation of a Bioactive Calcium-Phosphosilicate Bone Graft Material in Post-Extraction Alveloar Sockets. Gonshor A, Lanka M, Saroff S et.al., JIACD Dec 2011; 3(7): 21- 31.
8. Socket grafting with calcium phosphosilicate alloplast putty: A histomorphometric evaluation. Salama MA, Lanka M, Kurtzman GM, Joachim FPC, Compend Contin Educ Dent.. Sept 2012; 33(8): 109-115.
9. Practical application of the newly introduced natural bone regeneration (NBR) concept utilizing alloplastic putty. Kotsakis G, Chrepa V, Katta S Int. J Oral Imp Clin Res Sept-Dec 2011; 2(3):145-149.
10. Ridge preservation with a calcium phosphosilicate putty in 12 consecutive cases.. Kotsakis G, Chrepa V, Katta S., Clin Oral Implants Res. Sept 2011; 22(9):10-24.
GBR / Ridge Augmentation
1. Management of fenestration defects during flapless immediate implant placement in esthetic zone. Kher U, Tunkiwala A, Umrigar Z., Int J Periodontics Restorative Dent. Nov-Dec 2015; 35(6):e90-6.
2. Guided bone regeneration using titanium mesh with calcium phosphosilicate alloplast in the esthetic zone: A case report. Lanka M, Lahori M, Nagrath R, Sudan R., Int J Ultroson Piezoelect Bone Surg June 2014; 3(1): 19-24.
3. Clinical results of implant placement in resorbed ridges using simultaneous guided bone regeneration: a multicenter case series. Konstantinidis I, Kumar T, Kher U, Stanitsas PD, Hinrichs JE, Kotsakis GA., Clin Oral Investig. 2015 Mar;19(2):553-9.
4. Removal of a large radicular cyst and simultaneous ridge regeneration utilizing alloplastic CPS putty in combination with platelet rich fibrin (PRF): A case report. Kotsakis G, Chrepa V., JIACD Apr 2012; 4(2): 41-47.
5. Extraction of an impacted maxillary canine with immediate implant placement. Salama M, Lanka M, Kurtzman G., Inside Dentistry Mar 2012: 64-67.
Perio Defects
1. Clinical and cone beam computed tomography comparison of NovaBone Dental Putty and PerioGlas in the treatment of mandibular class II furcations. Asmita, Gupta V, Bains VK, Singh GP, Jhingran R., Indian J Dent Res, 2014 25(2):166-73.
2. Use of the NovaBone augmentation material in the treatment of chronic periodontitis. Preliminary communication. Slezak R, Paulusova V., ACTA MEDICA (Hradec Kralove) 2013:56(4):157-161.
3. Evaluation of the efficacy of bioactive synthetic graft material in the treatment of intrabony periodontal defects. Grover V, Kapoor A, Malhotra R, Uppal RS, J Indian Soc Periodontol. Jan-Feb 2013; 17(1): 104-110.
4. Effect of Addition of platelet-rich plasma to calcium phosphosilicate putty on healing at 9 months in periodontal intrabony defects. Shukla S, Chug, A, Mahesh L, Grover, HS, The J of Contemp Dent Prac Mar 2016; 17(3):230-234.
5. Comparative evaluation of bioactive glass putty and platelet rich fibrin in the treatment of human periodontal intrabony defects: a randomized control trial. Naqvi A, Gopalakrishnan D, Bhasin MT, Sharma, N, Haider K, Martande S. J of Clinical and Diagnostic Research 2017 Jul, Vol-11(7): ZC09-ZC13.
6. Comparative evaluation of bioactive glass (putty) and platelet rich fibrin in treating furcation defects. Biswas S, Sambashivaiah S, Kulal R, Bilichodmath S, Kurtzman GM. Journal of Oral Implantology Vol 17(5): 411-415
7. Comparative evaluation of bioactive synthetic novabone putty and calcified algae-derived porous hydroxyapatite bone grafts for the treatment of intrabony defects. Bembi NN, Bembi, S, Mago J, Baweja GK, Baweja PS. Int. J of Clin Pediatric Dent Oct-Dec 2016; 9(4):285-290.
8. Comparative evaluation of coronally advanced flap with and without bioactive glass putty in the management of gingival recession defects: a randomized controlled clinical trial. Bansal A, Kulloli A, Kathariya R, Shetty S, Jain H, Raiker S. J of Int. Academy of Perio 2016 18(1):7-15.
Published Clinical Cases
1. A simplified approach to the minimally invasive antral membrane elevation technique utilizing a viscoelastic medium for hydraulic sinus floor elevation. Kotsakis G., Mazor Z. Oral Maxillofac Surg. 2015 Mar; 19(1):97-101.
2. Bioactive glass in Periodontal Surgery and Implant Dentistry. Profeta AC, Prucher GM. Dental Materials Journal 2015; 34(5):559-571
3. Topical simvastatin improves the pro-angiogenic and pro-osteogenic properties of bioglass putty in the rat calvaria critical size model. Allon I, Anavi Y, Allon D. J Oral Implantology 2014; 40(3): 251-258
4. Synthetic Putty and Simultaneous Short Implant Placement in Crestal Sinus Lifting Procedures: 13–36 Months Follow-Up: A Case Series. Boogaard M. J Dent Rep. 2021; 2(1):1-8.
Product Guides
Text Books
- Bone Augmentation in Implant Dentistry - Michael A. Pikos, DDS
- Next-Generation Bio Materials for Bone & Periodontal Regeneration - By Richard J. Miron and Yufeng Zhang
SIZES & AVAILABILITY
NovaBone Dental Putty is available in multiple delivery options: trays, pre-filled syringes and a unique cartridge delivery system. The diameter of the cannula is 2.8mm which is ideal for dispensing of the graft, especially in minimally invasive techniques such as gaps in immediate implant and crestal approach sinus lifts. Cartridges are available in various sizes and are used in conjunction with cartridge dispenser.
Products are available through a network of distributors world-wide, with a presence in over 40 countries. Not all dispensing formats are available in all countries.
Please check with your local distributor for sizes, part # and availability.
Cartridge System
- 0.5cc Cartridges (blue) 2/Pack
- 0.5cc Cartridges (blue) 4/Pack
- 0.25cc Cartridges (green) 4/pack
- Cartridge dispensing gun
Syringe
- 0.5cc Syringe 1/Pack
- 0.5cc Syringe 2/Pack
- 1.0cc Syringe 1/Pack
- 2.0cc Syringe 1/Pack
Clam Shell
- 0.5cc Shell 1/Pack
- 0.5cc Shell 2/Pack
WHERE TO BUY
Our products are available through a network of distributors worldwide
Please contact your local dealer to learn more about NovaBone Products, local training events and specials offers
Learn more